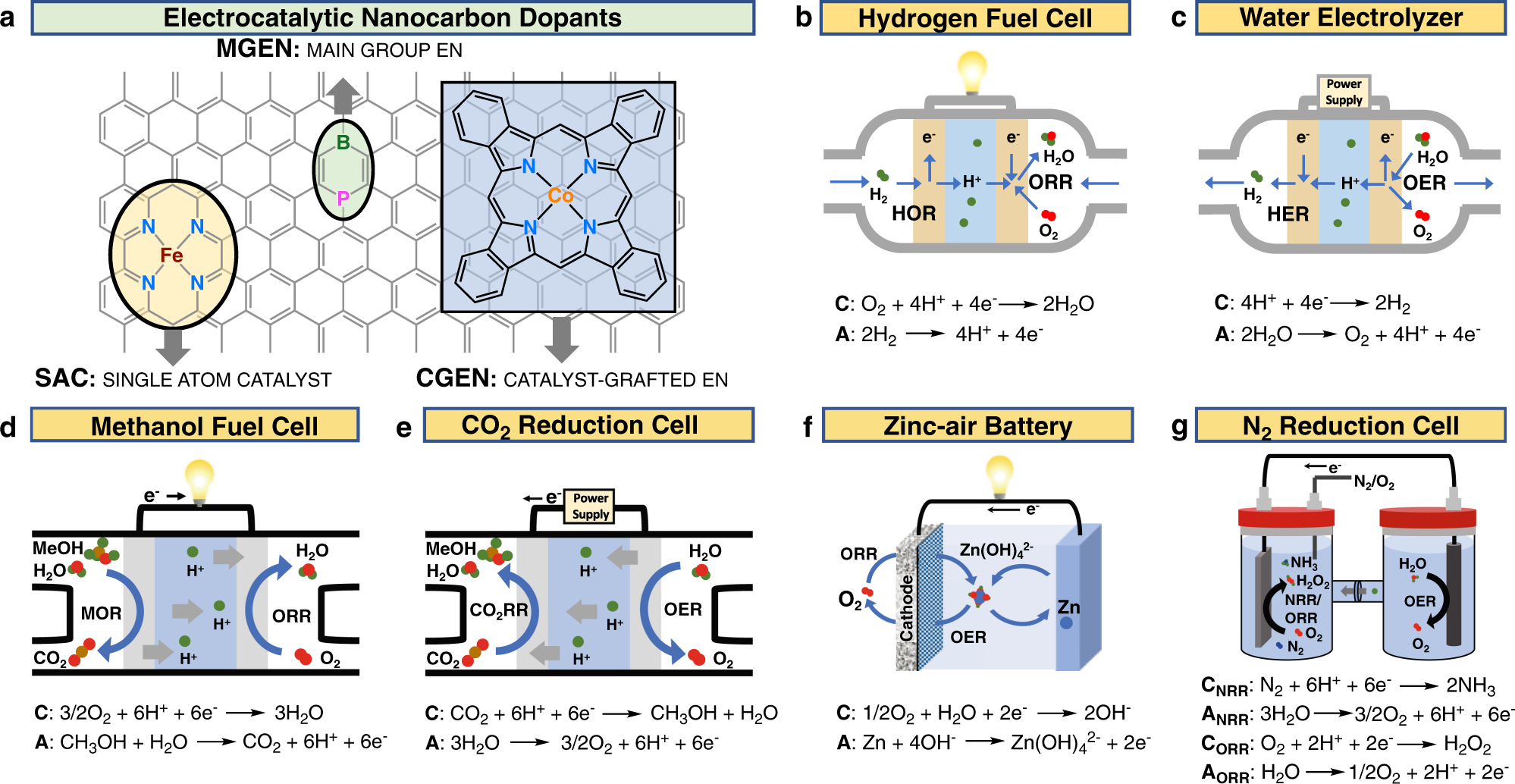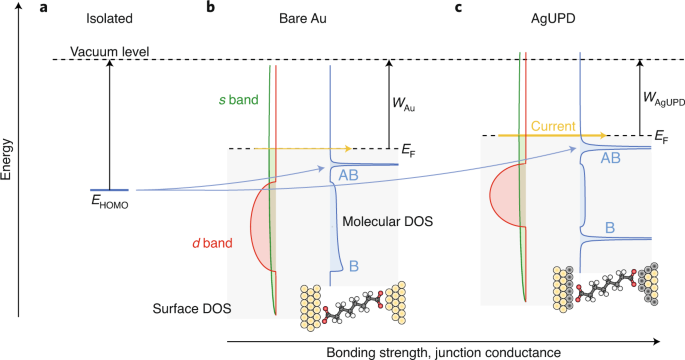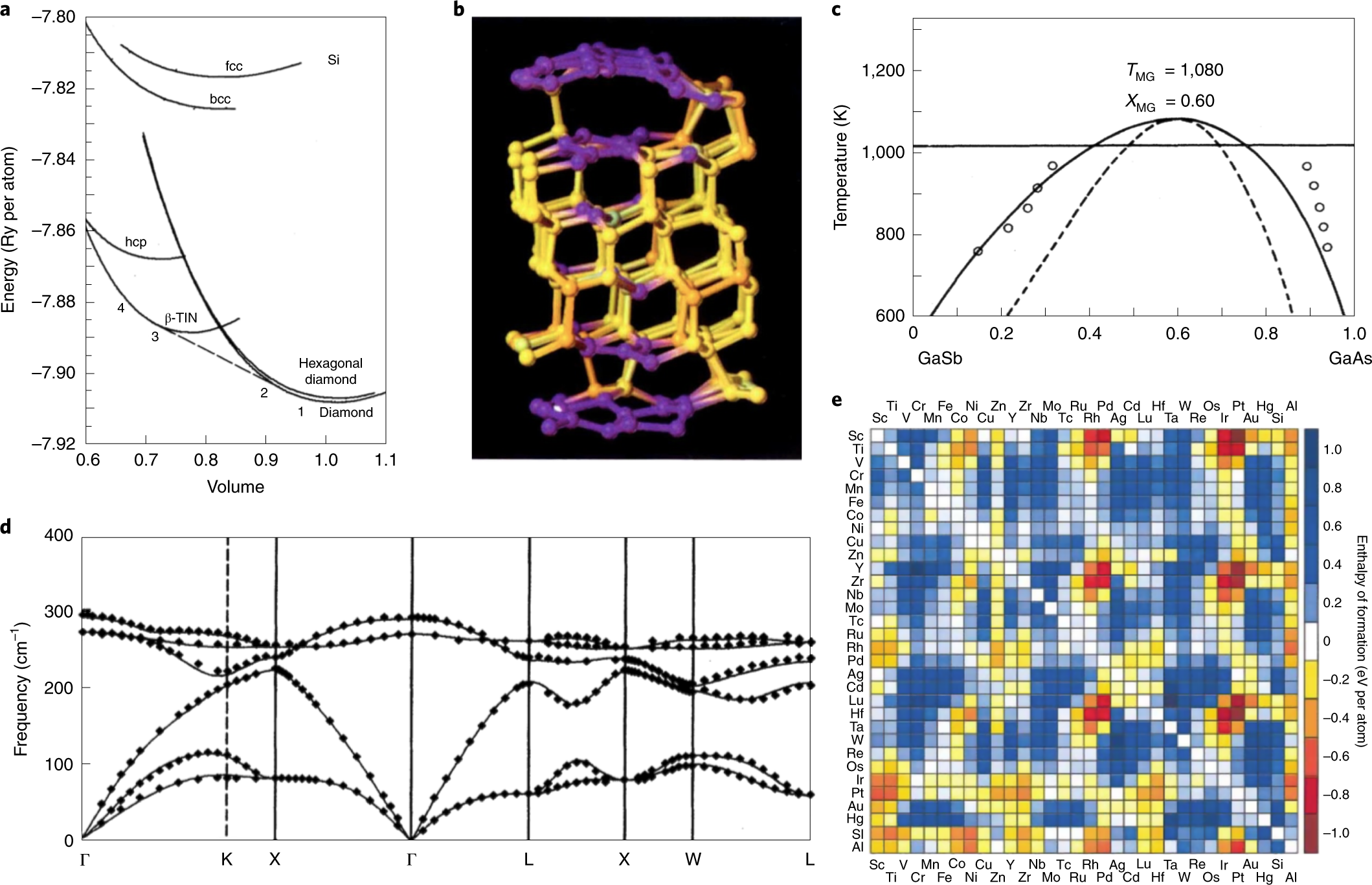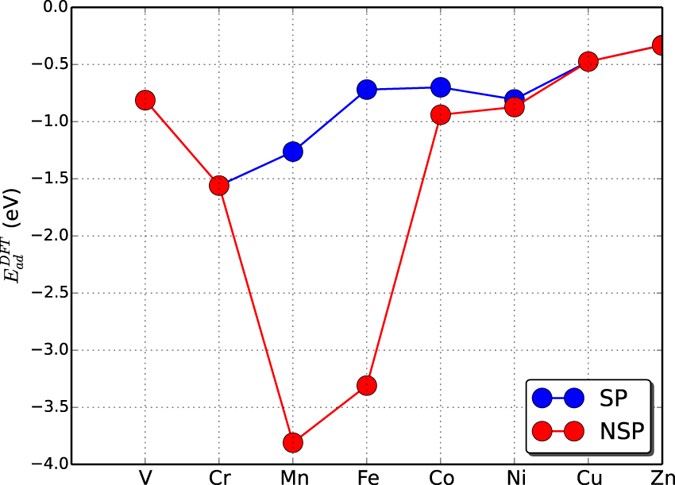
An improved d-band model of the catalytic activity of magnetic transition metal surfaces | Scientific Reports

Substrate doping: A strategy for enhancing reactivity on gold nanocatalysts by tuning sp bands: The Journal of Chemical Physics: Vol 143, No 14
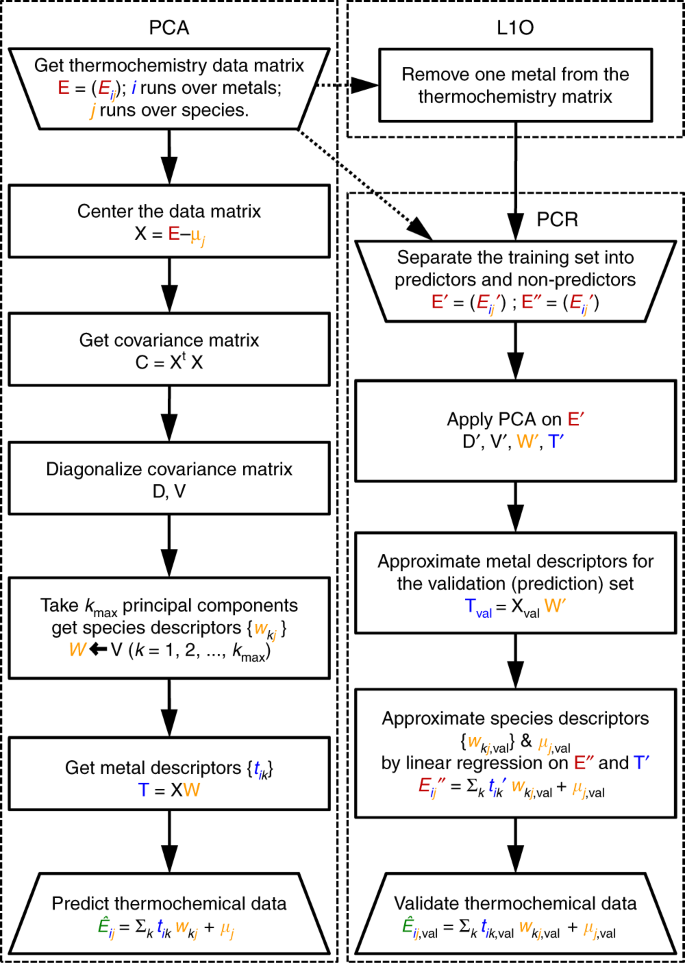
Statistical learning goes beyond the d-band model providing the thermochemistry of adsorbates on transition metals | Nature Communications

Dynamics of H2 dissociation on the close-packed (111) surface of the noblest metal: H2 + Au(111): The Journal of Chemical Physics: Vol 145, No 14
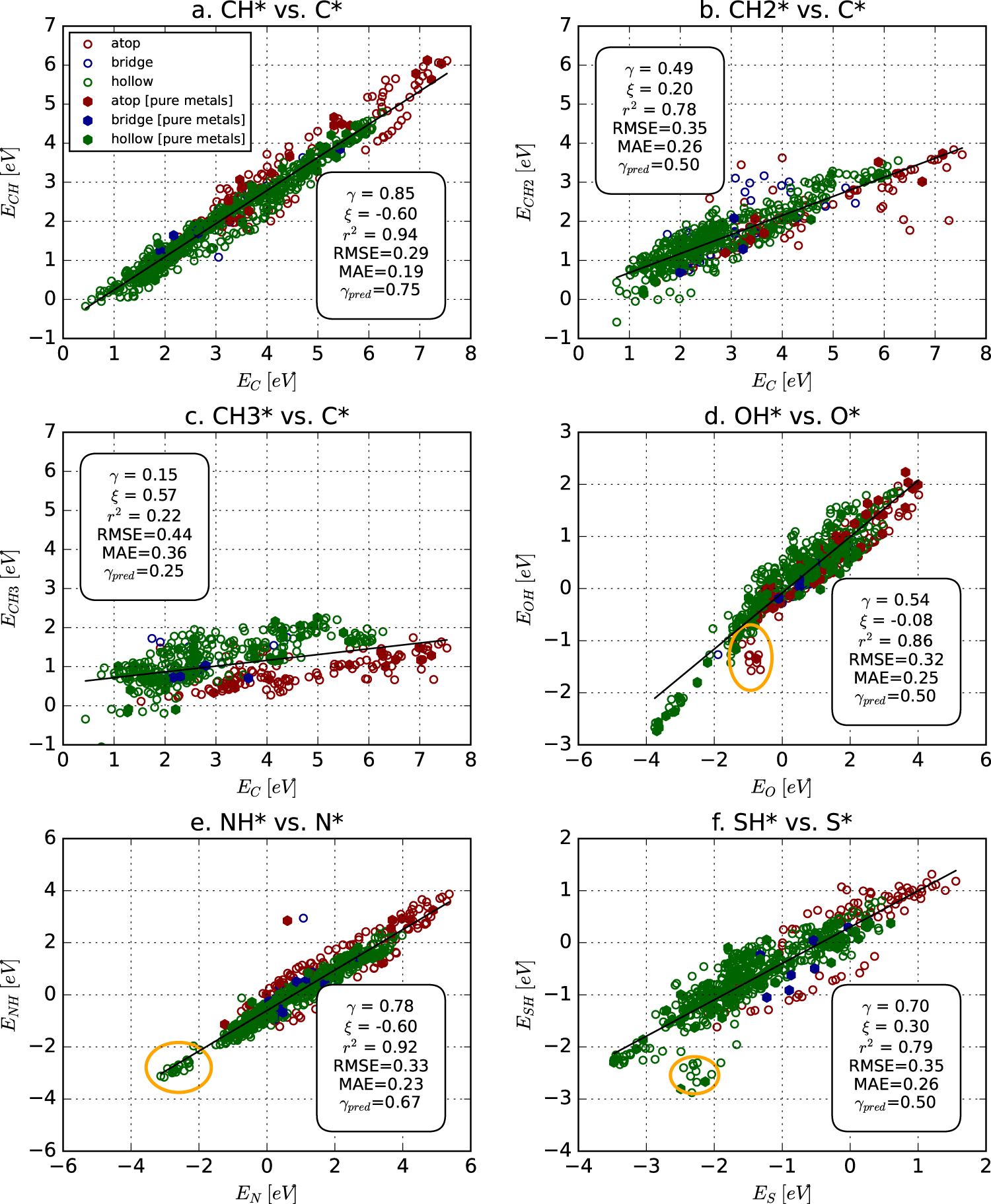
A Bayesian framework for adsorption energy prediction on bimetallic alloy catalysts | npj Computational Materials
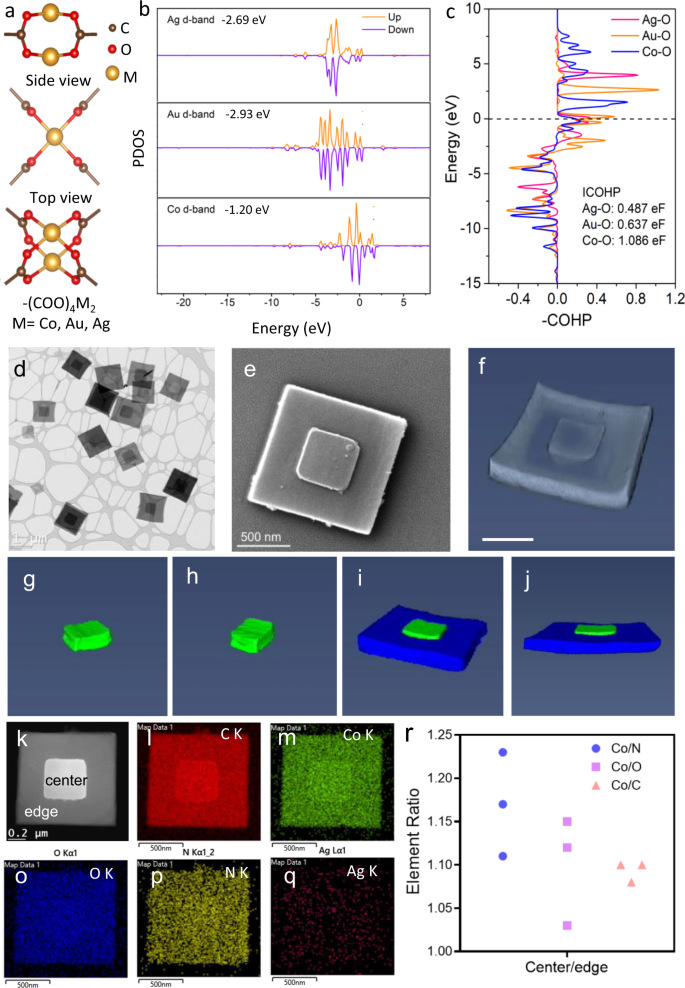
Phase-enabled metal-organic framework homojunction for highly selective CO2 photoreduction | Nature Communications
Characterization of methoxy adsorption on some transition metals: A first principles density functional theory study: The Journal of Chemical Physics: Vol 122, No 4

Communications: Exceptions to the d-band model of chemisorption on metal surfaces: The dominant role of repulsion between adsorbate states and metal d-states: The Journal of Chemical Physics: Vol 132, No 22

The perturbation energy: A missing key to understand the “nobleness” of bulk gold: The Journal of Chemical Physics: Vol 142, No 19

Communications: Exceptions to the d-band model of chemisorption on metal surfaces: The dominant role of repulsion between adsorbate states and metal d-states: The Journal of Chemical Physics: Vol 132, No 22

Directing reaction pathways via in situ control of active site geometries in PdAu single-atom alloy catalysts | Nature Communications

The density of states of graphene underneath a metal electrode and its correlation with the contact resistivity: Applied Physics Letters: Vol 103, No 3

The perturbation energy: A missing key to understand the “nobleness” of bulk gold: The Journal of Chemical Physics: Vol 142, No 19

Communications: Developing relationships between the local chemical reactivity of alloy catalysts and physical characteristics of constituent metal elements: The Journal of Chemical Physics: Vol 132, No 11

The perturbation energy: A missing key to understand the “nobleness” of bulk gold: The Journal of Chemical Physics: Vol 142, No 19

Wetting Induced Oxidation of Pt-based Nano Catalysts Revealed by In Situ High Energy Resolution X-ray Absorption Spectroscopy | Scientific Reports

Main Descriptors To Correlate Structures with the Performances of Electrocatalysts - Wang - - Angewandte Chemie International Edition - Wiley Online Library

Dynamics of H2 dissociation on the close-packed (111) surface of the noblest metal: H2 + Au(111): The Journal of Chemical Physics: Vol 145, No 14

Dynamics of H2 dissociation on the close-packed (111) surface of the noblest metal: H2 + Au(111): The Journal of Chemical Physics: Vol 145, No 14

Hydrogen induced lattice expansion and crystallinity degradation in palladium nanoparticles: Effect of hydrogen concentration, pressure, and temperature: Journal of Applied Physics: Vol 106, No 9

Single‐Crystal Cobalt Phosphate Nanosheets for Biomimetic Oxygen Evolution in Neutral Electrolytes - Shao - 2019 - Angewandte Chemie International Edition - Wiley Online Library